
Scientists working with data from NASA’s Cassini mission have confirmed the presence of a population of complex hydrocarbons in the upper atmosphere of Saturn’s largest moon, Titan, that later evolve into the components that give the moon a distinctive orange-brown haze. The presence of these complex, ringed hydrocarbons, known as polycyclic aromatic hydrocarbons (PAHs), explains the origin of the aerosol particles found in the lowest haze layer that blankets Titan’s surface. Scientists think these PAH compounds aggregate into larger particles as they drift downward.
“With the huge amount of methane in its atmosphere, Titan smog is like L.A. smog on steroids.”
– Scott Edgington, Cassini deputy project scientist
“With the huge amount of methane in its atmosphere, Titan smog is like L.A. smog on steroids,” said Scott Edgington, Cassini deputy project scientist based at NASA’s Jet Propulsion Laboratory, Pasadena, Calif. “These new papers using Cassini data shed light on how the heavy, complex hydrocarbon molecules that make up Titan’s smog came to form out of the simpler molecules in the atmosphere. Now that they have been identified, the longevity of Cassini’s mission will make it possible to study their variation with Titan seasons.”
Of all the bodies in the solar system, Saturn’s largest moon, Titan, has the atmosphere most resembling that of Earth. Like that of our planet, Titan’s atmosphere is largely composed of molecular nitrogen. Unlike Earth’s atmosphere, however, Titan’s contains only small traces of oxygen and water. Another molecule, methane, plays a similar role to that of water in Earth’s atmosphere, and makes up about 2 percent of Titan’s atmosphere. Scientists have speculated that the atmosphere of this moon may resemble that of our planet in its early days, before primitive living organisms enriched it with oxygen via photosynthesis.
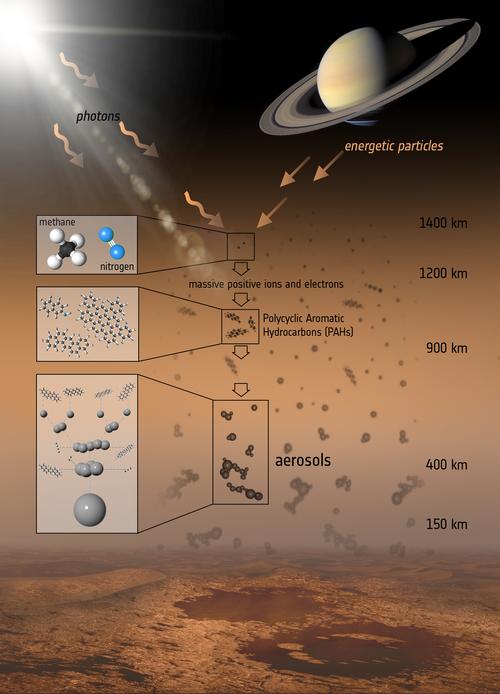
When sunlight or highly energetic particles from Saturn’s magnetic bubble hit the layers of Titan’s atmosphere above about 600 miles (1,000 kilometers), the nitrogen and methane molecules there are broken up. This results in the formation of massive positive ions and electrons, which trigger a chain of chemical reactions, producing a variety of hydrocarbons –- a wide range of which have been detected in Titan’s atmosphere. These reactions eventually lead to the production of carbon-based aerosols, large aggregates of atoms and molecules that are found in the lower layers of the haze that enshrouds Titan, well below 300 miles (500 kilometers)
The process is similar to Earth, where smog starts with sunlight breaking up hydrocarbons that are emitted into the air. The resulting pieces recombine to form more complex molecules.
“We can finally confirm that PAHs play a major role in the production of Titan’s lower haze, and that the chemical reactions leading to the formation of the haze start high up in the atmosphere,” said this paper’s lead author Manuel López-Puertas from the Astrophysics Institute of Andalucia in Granada, Spain. “This finding is surprising: we had long suspected that PAHs and aerosols were linked in Titan’s atmosphere, but didn’t expect we could prove this with current instruments.”
Reblogged this on Spherical Chickens in a Vacuum.
LikeLike
It’s very interesting new. So the the chemistry seems to work similar that on Earth. The only difference it is because on Titan it is some methane instead of the water…
Jeff Barani from Vence (France)
LikeLike
Yes, Titan has a hydrologic cycle, except with methane and ethane instead of H2O.
LikeLike